 This information on this page is strictly informative in nature and does not give users any rights. To obtain the of an atom, we start by counting the number of valence electrons for the neutral atom, and then subtract from it the number of electrons that it owns ( i.e. For the Lewis structure for PO4 3- you should take formal charges into account to find the best Lewis structure for the molecule. These exchange rates are available in electronic format from March 1994 in the form of downloadable files. 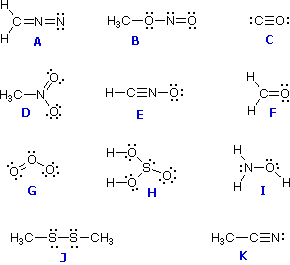
For structure I Phosphorus: formal charge5050 Oxygen: formal charge642 0 For structure II. 
For each currency, the converter provides the historic rates of conversion against the euro (or, until December 1998, against the ecu). We begin by calculating the formal charges. InforEuro provides rates for current and old currencies for countries both inside and outside the European Union. The rates indicated are the market rates for the second to last day of the previous month as quoted by the European Central Bank or, depending on availability, provided by the delegations or other appropriate sources close to that date. The converter also allows general users to get monthly currency conversion rates, from the current month back to 1994. Formal charges should be as small as possible (comparing the absolute value. A formal charge is defined as: The individual charge of each atom present in a molecule ADVERTISEMENT Determining the formal charge is of great significance. The formal charge on the NO2+ molecules oxygen terminal atoms often corresponds to the actual. The formula for calculating formal charge is: Fc Ve (B + Nb) F c V e ( B + N b), where Fc F c is the formal charge, Ve V e is the valence electrons in an isolated atom, B B is the number of bonds attached to the atom, and Nb N b is nonbonding electrons on the atom in the molecule. The sum of the formal charges must equal to the total charge on the molecule or ion. at the request of the European Commission. Calculating formal charge on the oxygen atom of NO2+ molecule. 
These rates are used to calculate amounts for the reimbursement of expenses, travel or subsistence costs for external people participating in meetings, interviews etc. Half of the number of bonded electrons For example: if an Oxygen atom in a molecule has a double bond and two lone pairs of electrons, its formal charge. This static currency converter provides the European Commission’s official monthly accounting rate for the euro and the conversion rates as established by the Accounting Officer of the European Commission in line with article 19 of the Financial Regulation. Apart from the formula, the formal charge can be calculated pictorially using the diagrammatic method. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
